As the microelectronic revolution changed the way how electronic components were manufactured 50 years ago, a similar development can be seen in the Life Sciences with the concept of the so-called “Lab-on-a-Chip” or microfluidics technology, which deals with the handling and manipulation of miniature amounts of liquids and was introduced almost 30 years ago [1]. After the number of scientific publications within the microfluidics area has dramatically increased between 2000 and 2010, the commercialization of microfluidics-enabled products has been picking up speed. We have seen the technology making a tremendous step from being a “technology looking for a problem” to a widely used truly enabling technology. Nowadays almost no product development in the field of diagnostics or analytical sciences takes place which does not involve elements with microfluidic functionality.
Several drivers behind the current commercial development can be named: Firstly, the fundamental scaling laws which favor miniaturization with mechanisms like diffusion and heat transport. This reduces overall time from the input of a sample to the analytical result to minutes rather than the hours or even days in larger systems. Secondly, the cost and the overall available volume of reagents in the Life Sciences is often a critical factor. By reducing these volumes, not only a cost reduction can be achieved but often this represents the only way of processing scarce material. Thirdly, many functional elements of biology, e.g. cells, blood vessels, bacteria etc. have a size which lies exactly in the range of microfabrication methods, making it an ideal fit between manufacturing technologies and applications. Fourthly, the very high geometrical accuracies of miniaturized systems together with the high surface-to-volume ratio makes the environment in which the fluids are contained extremely well controlled. Last but not least, miniaturization offers the potential to automate many laborious laboratory processes which often include many manual steps like pipetting, sample transfer etc., again reducing the cost and time of the complete analytical process and reducing the risk of procedural error. These advantages have proven to be very attractive, first spurning the very large scientific activity in the field and increasingly also in form of commercial products.
- Functional integration
One of the most important advances in recent years is the ability to transfer complex analytical or diagnostic processes onto a single microfluidics device.
Figure 1 shows typical process steps which have to be realized during a development of such an integrated microfluidic device.
In the first step, the sample has to be brought onto the device through some interface. As the type of sample can be very different (e.g. biopsy, swab, sputum, blood etc.), this interface has to be adapted to the type of sample. These “world-to-chip” interfaces still are an often over-looked item during the development.
The next step, the various sample preparation processes like liquefaction of the sample, the lysis of cells, extraction of DNA/RNA, the sample concentration etc., have so far been typically been carried out off-chip due to their complexity. Moving these steps onto the device represents the biggest challenge. Furthermore, many of these steps have to be carried out with a high precision in terms of volume, times or sequence. It is therefore a specific requirement in the development of miniaturized assays that the assay should be as robust as possible. The next process step frequently involves an amplification of target molecules, using methods like conventional or isothermal polymerase chain reaction (PCR) in order to increase the number of target molecules to achieve better detection selectivity and sensitivity. This amplification step is then usually followed by a separation step like electrophoresis, chromatography, the use of capture probes or other filtration mechanisms in order to isolate the desired component or remove unwanted components from the mixture.
The final analytical step comprises the detection of the analyte of interest. While for many larger, lab-based systems, optical detection methods like fluorescence still act as a benchmark with respect to sensitivity, for portable systems, electrochemical analysis methods or various other sensor methods (e.g. surface acoustic waves (SAW), quartz crystal microbalance, thermal measurements) are becoming increasingly of interest. It should be noted that all the preceding process steps have to be matched to the selected detection method in order to generate the best results.
A minor but nevertheless important design step of an integrated device in diagnostics is the layout of a waste container system in order to retain all liquids used in the process on-chip. This is often necessary to avoid the contamination risk of the instrument and to prevent carry-over from one measurement to the next.
Once these individual functions have been verified with microfluidic modules, a stepwise integration into a single device then can take place. An example for such an integrated microfluidic cartridge for molecular diagnostics is shown in Fig. 2.
Microfluidics technology has made enormous progress in the last 25 years and has proven that it is a technology which is viable in the scientific as well as the commercial arena. Although the commercial development did not happen as fast as many people predicted 20 years ago, it is evident that microfluidics has turned into a crucial enabling technology for almost any product development in the Life Sciences The big killer application is still missing, but comparison with the market uptake of other high-tech applications shows that the current time-line is nothing extraordinary. The range of applications is extremely broad and even if it has not revolutionized the Life Sciences as many had hoped for, it has and is currently changing many established practices in these disciplines.
References
[1] Manz, A., Graber, N. and Widmer, M. (1990) Miniaturized total chemical analysis systems: a novel concept for chemical sensing. Sens Actuators B1, 244-248
Figure captions:
Fig 1. Schematic diagram of the typical process steps involved in a bio-analytical or diagnostic process flow in a microfluidic device.
Fig. 2: Integrated microfluidic device for molecular diagnostics.
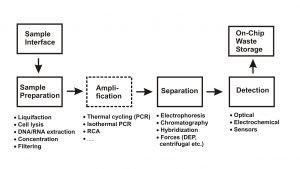
Fig. 1:
Fig 1. Schematic diagram of the typical process steps involved in a bio-analytical or diagnostic process flow in a microfluidic device.

Fig. 2: Integrated microfluidic device for molecular diagnostics.


